Top science and technology news from Madagascar
Provided by AGPCytokinetics Announces Positive Topline Results from ACACIA-HCM, the Pivotal Phase 3 Clinical Trial of Aficamten in Patients with Non-Obstructive Hypertrophic Cardiomyopathy
Trial Met Dual Primary Endpoints of KCCQ and Maximal Exercise Performance
With Consistent Positive Findings Across Key Secondary Endpoints
Company to Host Conference Call and Webcast Tuesday May 5 at 8:00 AM Eastern Time
SOUTH SAN FRANCISCO, Calif., May 05, 2026 (GLOBE NEWSWIRE) -- Cytokinetics, Incorporated (Nasdaq: CYTK) today announced positive topline results from ACACIA-HCM (Assessment Comparing Aficamten to Placebo on Cardiac Endpoints In Adults with Non-Obstructive HCM), the pivotal Phase 3 clinical trial of aficamten in patients with symptomatic non-obstructive hypertrophic cardiomyopathy (HCM).
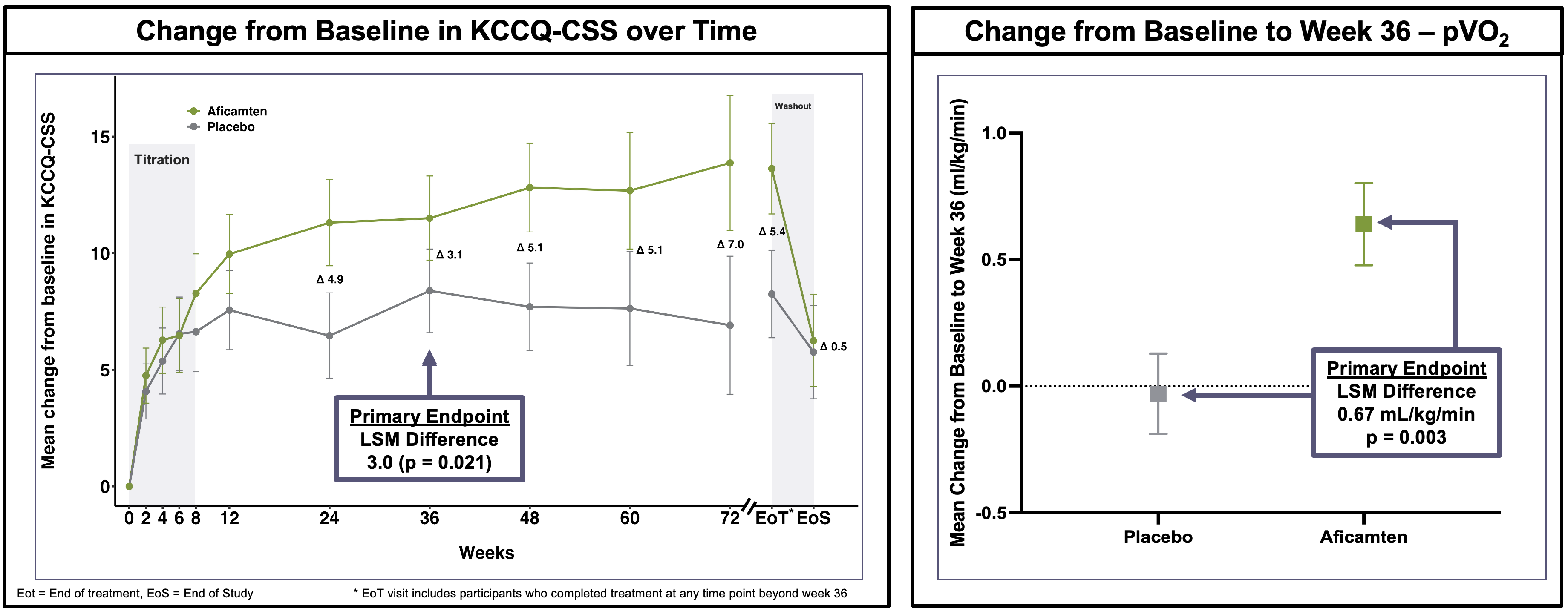
ACACIA-HCM met both dual primary endpoints, demonstrating statistically significant improvements from baseline to Week 36 compared to placebo in both Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and maximal exercise performance (pVO2) (Table 1).
“Patients with non-obstructive HCM have no therapies approved to treat the underlying hypercontractility associated with the disease. We hope that will change with ACACIA-HCM which is the first clinical trial to demonstrate statistically significant improvements in exercise capacity and symptom burden in patients with non-obstructive HCM,” said Fady I. Malik, M.D., Ph.D., Cytokinetics’ Executive Vice President of Research & Development. “We believe that the totality and consistency of evidence favoring aficamten across multiple patient-reported and physician-assessed endpoints of symptom improvement and physical function are clinically meaningful for patients with non-obstructive HCM.”
Table 1: Primary Endpoint Results
| Primary Endpoints |
Change from Baseline to Week 36 LSM (95% CI) |
Aficamten vs Placebo LSM (95% CI) |
p-value | |
| Aficamten | Placebo | |||
| KCCQ-CSS | 11.4 (9.6 – 13.2) | 8.4 (6.6 – 10.2) | 3.0 (0.5 - 5.5) | 0.021 |
| pVO2 (mL/kg/min) | 0.64 (0.32 – 0.95) | -0.03 (-0.35 – 0.28) | 0.67 (0.22 - 1.1) | 0.003 |
| LSM = least square mean; CI = confidence interval | ||||
The improvement in KCCQ was robust and consistent throughout the treatment period in participants on aficamten. Following washout, KCCQ decreased for participants on aficamten to match the placebo group. At Week 36, pVO2 increased for participants on aficamten, while it remained unchanged for participants on placebo, consistent with prior trials of aficamten in obstructive HCM (oHCM) (Figure 1).
Figure 1: Assessments of KCCQ and pVO2
Statistically significant (p<0.001) improvements compared to placebo were observed in key secondary endpoints including the proportion of participants with improvements in New York Heart Association (NYHA) Functional Class, the composite z-score of ventilatory efficiency and pVO2, and NT-proBNP.
There were no new safety signals identified. The percentage of participants completing planned dosing was similar in those receiving aficamten or placebo (88.4% vs. 90.3%, respectively). Left ventricular ejection fraction (LVEF) <50% occurred in 27 (10%) participants taking aficamten and in two (1%) participants taking placebo. Two participants on aficamten experienced a serious adverse event of heart failure associated with LVEF <50%. Treatment interruptions due to LVEF <40% occurred in 3% of participants taking aficamten.
“We are grateful to the clinical trial investigators and staff, as well as the patients who participated in this trial,” Dr. Malik added. “We look forward to presenting the results from ACACIA-HCM at an upcoming medical meeting, as well as discussing them with the U.S. FDA and other regulatory authorities.”
Investor Webcast Information
Cytokinetics will host an investor conference call on May 5, 2026, at 8:00 AM Eastern Time to discuss the topline results from ACACIA-HCM. Interested parties can register online at ACACIA-HCM Topline Results. The live webcast will be available on the Investors & Media section of the Cytokinetics website at https://ir.cytokinetics.com/. A replay of the webcast will be archived on the Cytokinetics website for six months.
About ACACIA-HCM
ACACIA-HCM was a Phase 3, multi-center, randomized, double-blind, placebo-controlled clinical trial designed to evaluate the effect of aficamten compared to placebo in patients with symptomatic non-obstructive hypertrophic cardiomyopathy (nHCM). The dual primary endpoint was the change in Kansas City Cardiomyopathy Questionnaire (KCCQ) Clinical Summary Score and change in maximal exercise performance (pVO2) from baseline to Week 36.
Secondary endpoints included the proportion of participants with ≥1 class improvement in New York Heart Association (NYHA) functional class, and changes in the composite z-score of two cardiopulmonary exercise testing (CPET) parameters of sub-maximal exercise performance (VE/VCO2 and pVO2), NT-proBNP, and left atrial volume index (LAVI) from baseline to Week 36. After 36 weeks of treatment, participants continued treatment with aficamten or placebo for up to 72 weeks to evaluate additional secondary and exploratory analyses including the time to first cardiovascular event. The trial (outside Japan) concluded when at least 200 participants completed 52 weeks of treatment.
ACACIA-HCM randomized and treated 516 participants (outside Japan) on a 1:1 basis with aficamten or placebo. Randomization was stratified by persistent atrial fibrillation and presence of intracavitary obstruction. At screening, participants enrolled in ACACIA-HCM were required to have resting left ventricular outflow tract gradient (LVOT-G) <30 mmHg and post-Valsalva LVOT-G <50 mmHg in addition to left ventricular ejection fraction (LVEF) ≥60%, respiratory exchange ratio (RER) ≥1.00 and peak VO2 ≤90% predicted, NT-proBNP ≥300 pg/mL or ≥900 pg/mL if atrial fibrillation or atrial flutter were present at screening, NYHA functional class II or III and KCCQ Clinical Summary Score ≤85.
Each patient received up to four escalating doses of aficamten or placebo based on echocardiographic guidance. Participants who received aficamten began with 5 mg dosed once daily. At weeks 2, 4 and 6 participants received an echocardiogram to determine if they would be up-titrated to escalating doses of 10, 15 or 20 mg. Dose escalation occurred only if a participant had an LVEF ≥60%. Participants who did not meet escalation criteria continued the same dose or were down-titrated if their LVEF was <50%.
About MYQORZO® (aficamten)
MYQORZO® (aficamten) is a cardiac myosin inhibitor approved in the U.S., China and European Union for the treatment of symptomatic obstructive hypertrophic cardiomyopathy (oHCM). In patients with oHCM, myosin inhibition with MYQORZO reduces cardiac contractility and consequently, left ventricular outflow tract (LVOT) obstruction. MYQORZO was engineered to achieve a predictable exposure response, rapid onset of action and reversibility.1
Aficamten is also under clinical investigation in CEDAR-HCM, in a pediatric population with oHCM. Aficamten has not been deemed safe or effective for use in this patient population. In addition, aficamten is being studied in FOREST-HCM, an open-label extension clinical study.
INDICATION
MYQORZO is indicated for the treatment of adults with symptomatic obstructive hypertrophic cardiomyopathy (oHCM) to improve functional capacity and symptoms.
IMPORTANT SAFETY INFORMATION
|
WARNING: RISK OF HEART FAILURE MYQORZO reduces left ventricular ejection fraction (LVEF) and can cause heart failure due to systolic dysfunction. Echocardiogram assessments are required prior to and during treatment with MYQORZO to monitor for systolic dysfunction. Initiation of MYQORZO in patients with LVEF <55% is not recommended. Decrease the dose of MYQORZO if LVEF is <50% and ≥40%. Interrupt the dose of MYQORZO if LVEF <40% or if the patient experiences heart failure symptoms or worsening clinical status due to systolic dysfunction. Because of the risk of heart failure due to systolic dysfunction, MYQORZO is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the MYQORZO REMS Program. |
CONTRAINDICATIONS
MYQORZO is contraindicated with concomitant use of rifampin.
WARNINGS AND PRECAUTIONS
Heart Failure
MYQORZO reduces cardiac contractility, which can reduce LVEF and cause heart failure.
Patients who experience a serious intercurrent illness (eg, serious infection) or arrhythmia (eg, new or uncontrolled atrial fibrillation) may be at greater risk of developing systolic dysfunction and heart failure.
Assess patients’ clinical status and LVEF prior to and during treatment and adjust the MYQORZO dose accordingly. New or worsening arrhythmia, dyspnea, chest pain, fatigue, leg edema, or elevations in N-terminal pro-B-type natriuretic peptide may be signs and symptoms of heart failure.
Initiation of MYQORZO in patients with LVEF <55% is not recommended.
MYQORZO REMS Program
MYQORZO is available only through a restricted program called the MYQORZO REMS Program, because of the risk of heart failure due to systolic dysfunction.
Notable requirements of the MYQORZO REMS Program include:
- Prescribers must be certified by enrolling in the MYQORZO REMS Program
- Patients must enroll in the MYQORZO REMS Program and comply with ongoing monitoring requirements
- Pharmacies must be certified by enrolling in the MYQORZO REMS Program and must only dispense to patients who are authorized to receive MYQORZO
- Wholesalers and distributors must only distribute to certified pharmacies
Further information is available at www.MYQORZOREMS.com, or at 1-844-285-7367.
Cytochrome P450 Interactions Leading to Heart Failure or Loss of Effectiveness
MYQORZO is metabolized primarily by CYP2C9, and to a lesser extent by CYP3A, CYP2D6, and CYP2C19 enzymes. Initiation of medications that inhibit multiple P450 pathways of MYQORZO elimination (eg, fluconazole, voriconazole, or fluvoxamine) or strong CYP2C9 inhibitors, and discontinuation of moderate-to-strong CYP3A inducers may lead to increased blood concentrations of aficamten and increase the risk of heart failure due to systolic dysfunction. Conversely, initiation of medications that induce P450 pathways of MYQORZO (eg, rifampin, moderate-to-strong CYP3A inducers) may lead to decreased blood concentrations of aficamten and potential loss of effectiveness. Assess LVEF 2 to 8 weeks after initiation of such inhibitors or after discontinuation of such inducers and adjust the dose of MYQORZO accordingly.
Advise patients of the potential for drug interactions. Advise patients to inform their healthcare provider of all concomitant medications prior to and during MYQORZO treatment.
ADVERSE REACTIONS
Hypertension (8% vs 2%) was the only adverse reaction occurring in >5% of patients and more commonly on MYQORZO than on placebo in the pivotal trial.
INDICATIONS AND USAGE
MYQORZO is indicated for the treatment of adults with symptomatic obstructive hypertrophic cardiomyopathy (oHCM) to improve functional capacity and symptoms.
Please see full Prescribing Information, including Boxed WARNING.
About Hypertrophic Cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is a disease in which the heart muscle becomes abnormally thick. HCM can be obstructive, when thickened muscle blocks blood flow, or non-obstructive, when blood flow is not blocked but heart function is still affected. In obstructive HCM, the thickening of cardiac muscle leads to the inside of the left ventricle becoming smaller, stiffer and less able to relax and fill with blood. Ultimately, HCM limits the heart’s pumping function, leading to reduced exercise capacity and a variety of symptoms.
HCM is the most common monogenic inherited cardiovascular disorder, with well over 300,000 patients diagnosed in the U.S. However, there are an estimated 400,000-800,000 additional patients who remain undiagnosed.2,3,4 Recent analysis of a large claims database indicates that approximately half of patients with HCM have obstructive HCM (oHCM) and half have non-obstructive HCM (nHCM).5
People with HCM are at high risk of also developing cardiovascular complications including atrial fibrillation, stroke and mitral valve disease.6 People with HCM are at risk for potentially fatal ventricular arrhythmias and it is one of the leading causes of sudden cardiac death in younger people or athletes.7 A subset of patients with HCM are at high risk of progressive disease leading to dilated cardiomyopathy and heart failure necessitating cardiac transplantation. There are no currently approved therapies for nHCM.
About Cytokinetics
Cytokinetics is a specialty cardiovascular biopharmaceutical company, building on its over 25 years of pioneering scientific innovations in muscle biology, and advancing a pipeline of potential new medicines for patients suffering from diseases of cardiac muscle dysfunction. Cytokinetics’ MYQORZO® (aficamten) is a cardiac myosin inhibitor approved in the U.S., European Union and China for the treatment of adults with symptomatic obstructive hypertrophic cardiomyopathy (oHCM). Cytokinetics is also developing omecamtiv mecarbil, an investigational cardiac myosin activator for the potential treatment of patients with heart failure with severely reduced ejection fraction and ulacamten, an investigational cardiac myosin inhibitor for the potential treatment of heart failure with preserved ejection fraction, while continuing pre-clinical research and development in muscle biology.
For additional information about Cytokinetics, visit www.cytokinetics.com and follow us on X, LinkedIn, Facebook and YouTube.
Forward-Looking Statements
This press release contains forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995 (the “Act”). Cytokinetics disclaims any intent or obligation to update these forward-looking statements and claims the protection of the Act’s Safe Harbor for forward-looking statements. Examples of such statements include, but are not limited to, statements relating to the enrollment, expected results or timing of completion of any of our clinical trials, the clinical meaningfulness, persuasiveness or interpretation of clinical trial results, including for purposes of regulatory approval, labeling, or market acceptance, the results of long-term, secondary or exploratory analyses, including analyses of time to first cardiovascular event, statements relating to our ability to obtain regulatory approval for aficamten in nonobstructive hypertrophic cardiomyopathy in any jurisdiction by any particular date, if ever, the number of patients comprising the eligible treatment population for aficamten, or market acceptance of aficamten for the treatment of nonobstructive hypertrophic cardiomyopathy. Such statements are based on management’s current expectations, but actual results may differ materially due to various risks and uncertainties, including, but not limited to, potential difficulties or delays in the development, testing, regulatory approvals for trial commencement, progression or product sale or manufacturing of Cytokinetics’ drug candidates that could slow or prevent clinical development or product approval; Cytokinetics’ drug candidates may have adverse side effects or inadequate therapeutic efficacy; the FDA or foreign regulatory agencies may delay or limit Cytokinetics’ ability to conduct clinical trials; Cytokinetics may be unable to obtain or maintain patent or trade secret protection for its intellectual property; standards of care may change, rendering Cytokinetics’ drug candidates obsolete; and competitive products or alternative therapies may be developed by others for the treatment of indications Cytokinetics’ drug candidates and potential drug candidates may target. For further information regarding these and other risks related to Cytokinetics’ business, investors should consult Cytokinetics’ filings with the Securities and Exchange Commission including the risk factors included in Cytokinetics’ most recent Annual Report on Form 10-K and subsequent reports filed with the SEC.
CYTOKINETICS® and the CYTOKINETICS C-shaped logo are registered trademarks of Cytokinetics in the U.S. and certain other countries.
MYQORZO® is a registered trademark of Cytokinetics in the U.S. and the European Union.
References
- Hartman JJ, Hwee DT, Robert-Paganin J, et al. Aficamten is a small-molecule cardiac myosin inhibitor designed to treat hypertrophic cardiomyopathy. Nat Cardiovasc Res. 2024;3(8) :1003-1016. doi:10.1038/s44161-024-00505-0
- CVrg: Heart Failure 2020-2029, p 44; Maron et al. 2013 doi:10.1016/S0140-6736(12)60397-3; Maron et al 2018 10.1056/NEJMra1710575
- Symphony Health 2016-2021 Patient Claims Data DoF;
- Maron MS, Hellawell JL, Lucove JC, Farzaneh-Far R, Olivotto I. Occurrence of Clinically Diagnosed Hypertrophic Cardiomyopathy in the United States. Am J Cardiol. 2016; 15;117(10):1651-1654.
- Butzner M, et al. Epidemiology of Hypertrophic Cardiomyopathy in the United States From 2016 to 2023. JACC Adv. 2026. 2026;5(2):102552. doi:10.1016/j.jacadv.2025.102552
- Gersh, B.J., Maron, B.J., Bonow, R.O., Dearani, J.A., Fifer, M.A., Link, M.S., et al. 2011 ACCF/AHA guidelines for the diagnosis and treatment of hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Journal of the American College of Cardiology and Circulation, 58, e212-260.
- Hong Y, Su WW, Li X. Risk factors of sudden cardiac death in hypertrophic cardiomyopathy. Current Opinion in Cardiology. 2022 Jan 1;37(1):15-21
Contact:
Cytokinetics
Diane Weiser
Senior Vice President, Corporate Affairs
(415) 290-7757
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/adcf634e-a2a7-4cdf-98b3-ca658eab1b35

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.


